Intermittent low-level vagosympathetic nerve trunk stimulation inhibits ganglionated plexi activity to prevent atrial fibrillation.
Lu, Y; Sun, J; Zhang, L; Sun, Q; Zhou, X; Li, J; Zhang, Y; Tang, B
International journal of clinical and experimental medicine
8
5094-102
2015
Show Abstract
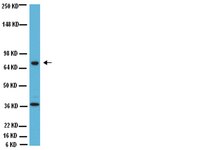
The cardiac autonomic nervous system (CANS) plays a role in the occurrence and persistence of atrial fibrillation (AF). Low-level vagosympathetic nerve stimulation (LL-VNS) has been shown to inhibit the occurrence of AF.The novel objective of this study was to compare the effects of intermittent low- level vagosympathetic nerve stimulation (I-VNS) and continuous low-level vagosympathetic nerve stimulation (C-VNS).19 beagles were randomly divided into 3 groups: Group A, rapid left atrial appendage pacing for 6 hours; Group B, rapid atrial pacing (RAP) for 6 hours and C-VNS (20 Hz, interval 0.1 ms, square wave) with 50% threshold voltage strength; Group C, RAP for 6 hours and I-VNS (continuously recurring cycles of 30-second ON, 30-second OFF). The atrial monophasic action potential (MAP) and the effective refractory periods (ERP) of the atrium and the pulmonary veins were measured at baseline, 1 hour, 3 hours and 6 hours after the experiment began. After the experiment, tyrosine hydroxylase (TH) and choline acetyl transferase (CHAT) expression levels in the anterior right ganglionated plexi (ARGP) from each group were measured.Inter-group comparisons of MAP and ERP demonstrated that Group A was significantly different from Groups B and C (P less than 0.05), while the difference between Groups B and C was not significant (P greater than 0.05). The MAP and ERP in Group A gradually decreased, reaching a minimum at 6 hours, but no significant changes were observed in Groups B and C. When compared to Group A, both Groups B and C had reduced TH and CHAT expression.During the occurrence and development of AF, I-VNS could protect the cardiovascular system, possibly replacing C-VNS. Additionally, both I-VNS and C-VNS inhibited ganglionated plexus (GP) activity during the AF prevention. | | | 26131082
 |
Distinctive receptive field and physiological properties of a wide-field amacrine cell in the macaque monkey retina.
Manookin, MB; Puller, C; Rieke, F; Neitz, J; Neitz, M
Journal of neurophysiology
114
1606-16
2015
Show Abstract
At early stages of visual processing, receptive fields are typically described as subtending local regions of space and thus performing computations on a narrow spatial scale. Nevertheless, stimulation well outside of the classical receptive field can exert clear and significant effects on visual processing. Given the distances over which they occur, the retinal mechanisms responsible for these long-range effects would certainly require signal propagation via active membrane properties. Here the physiology of a wide-field amacrine cell-the wiry cell-in macaque monkey retina is explored, revealing receptive fields that represent a striking departure from the classic structure. A single wiry cell integrates signals over wide regions of retina, 5-10 times larger than the classic receptive fields of most retinal ganglion cells. Wiry cells integrate signals over space much more effectively than predicted from passive signal propagation, and spatial integration is strongly attenuated during blockade of NMDA spikes but integration is insensitive to blockade of NaV channels with TTX. Thus these cells appear well suited for contributing to the long-range interactions of visual signals that characterize many aspects of visual perception. | | | 26133804
 |
Transmitter inputs to different motoneuron subgroups in the oculomotor and trochlear nucleus in monkey.
Zeeh, C; Mustari, MJ; Hess, BJ; Horn, AK
Frontiers in neuroanatomy
9
95
2015
Show Abstract
In all vertebrates the eyes are moved by six pairs of extraocular muscles enabling horizontal, vertical and rotatory movements. Recent work showed that each extraocular muscle is controlled by two motoneuronal groups: (1) Motoneurons of singly-innervated muscle fibers (SIF) that lie within the boundaries of motonuclei mediating a fast muscle contraction; and (2) motoneurons of multiply-innervated muscle fibers (MIF) in the periphery of motonuclei mediating a tonic muscle contraction. Currently only limited data about the transmitter inputs to the SIF and MIF motoneurons are available. Here we performed a quantitative study on the transmitter inputs to SIF and MIF motoneurons of individual muscles in the oculomotor and trochlear nucleus in monkey. Pre-labeled motoneurons were immunostained for GABA, glutamate decarboxylase, GABA-A receptor, glycine transporter 2, glycine receptor 1, and vesicular glutamate transporters 1 and 2. The main findings were: (1) the inhibitory control of SIF motoneurons for horizontal and vertical eye movements differs. Unlike in previous primate studies a considerable GABAergic input was found to all SIF motoneuronal groups, whereas a glycinergic input was confined to motoneurons of the medial rectus (MR) muscle mediating horizontal eye movements and to those of the levator palpebrae (LP) muscle elevating the upper eyelid. Whereas SIF and MIF motoneurons of individual eye muscles do not differ numerically in their GABAergic, glycinergic and vGlut2 input, vGlut1 containing terminals densely covered the supraoculomotor area (SOA) targeting MR MIF motoneurons. It is reasonable to assume that the vGlut1 input affects the near response system in the SOA, which houses the preganglionic neurons mediating pupillary constriction and accommodation and the MR MIF motoneurones involved in vergence. | | | 26257611
 |
Vibrissa Self-Motion and Touch Are Reliably Encoded along the Same Somatosensory Pathway from Brainstem through Thalamus.
Moore, JD; Mercer Lindsay, N; Deschênes, M; Kleinfeld, D
PLoS biology
13
e1002253
2015
Show Abstract
Active sensing involves the fusion of internally generated motor events with external sensation. For rodents, active somatosensation includes scanning the immediate environment with the mystacial vibrissae. In doing so, the vibrissae may touch an object at any angle in the whisk cycle. The representation of touch and vibrissa self-motion may in principle be encoded along separate pathways, or share a single pathway, from the periphery to cortex. Past studies established that the spike rates in neurons along the lemniscal pathway from receptors to cortex, which includes the principal trigeminal and ventral-posterior-medial thalamic nuclei, are substantially modulated by touch. In contrast, spike rates along the paralemniscal pathway, which includes the rostral spinal trigeminal interpolaris, posteromedial thalamic, and ventral zona incerta nuclei, are only weakly modulated by touch. Here we find that neurons along the lemniscal pathway robustly encode rhythmic whisking on a cycle-by-cycle basis, while encoding along the paralemniscal pathway is relatively poor. Thus, the representations of both touch and self-motion share one pathway. In fact, some individual neurons carry both signals, so that upstream neurons with a supralinear gain function could, in principle, demodulate these signals to recover the known decoding of touch as a function of vibrissa position in the whisk cycle. | | | 26393890
 |
Pharmacogenetic stimulation of cholinergic pedunculopontine neurons reverses motor deficits in a rat model of Parkinson's disease.
Pienaar, IS; Gartside, SE; Sharma, P; De Paola, V; Gretenkord, S; Withers, D; Elson, JL; Dexter, DT
Molecular neurodegeneration
10
47
2015
Show Abstract
Patients with advanced Parkinson's disease (PD) often present with axial symptoms, including postural- and gait difficulties that respond poorly to dopaminergic agents. Although deep brain stimulation (DBS) of a highly heterogeneous brain structure, the pedunculopontine nucleus (PPN), improves such symptoms, the underlying neuronal substrate responsible for the clinical benefits remains largely unknown, thus hampering optimization of DBS interventions. Choline acetyltransferase (ChAT)::Cre(+) transgenic rats were sham-lesioned or rendered parkinsonian through intranigral, unihemispheric stereotaxic administration of the ubiquitin-proteasomal system inhibitor, lactacystin, combined with designer receptors exclusively activated by designer drugs (DREADD), to activate the cholinergic neurons of the nucleus tegmenti pedunculopontine (PPTg), the rat equivalent of the human PPN. We have previously shown that the lactacystin rat model accurately reflects aspects of PD, including a partial loss of PPTg cholinergic neurons, similar to what is seen in the post-mortem brains of advanced PD patients.In this manuscript, we show that transient activation of the remaining PPTg cholinergic neurons in the lactacystin rat model of PD, via peripheral administration of the cognate DREADD ligand, clozapine-N-oxide (CNO), dramatically improved motor symptoms, as was assessed by behavioral tests that measured postural instability, gait, sensorimotor integration, forelimb akinesia and general motor activity. In vivo electrophysiological recordings revealed increased spiking activity of PPTg putative cholinergic neurons during CNO-induced activation. c-Fos expression in DREADD overexpressed ChAT-immunopositive (ChAT+) neurons of the PPTg was also increased by CNO administration, consistent with upregulated neuronal activation in this defined neuronal population.Overall, these findings provide evidence that functional modulation of PPN cholinergic neurons alleviates parkinsonian motor symptoms. | | | 26394842
 |
Rac1 plays an essential role in axon growth and guidance and in neuronal survival in the central and peripheral nervous systems.
Hua, ZL; Emiliani, FE; Nathans, J
Neural development
10
21
2015
Show Abstract
Rac1 is a critical regulator of cytoskeletal dynamics in multiple cell types. In the nervous system, it has been implicated in the control of cell proliferation, neuronal migration, and axon development.To systematically investigate the role of Rac1 in axon growth and guidance in the developing nervous system, we have examined the phenotypes associated with deleting Rac1 in the embryonic mouse forebrain, in cranial and spinal motor neurons, in cranial sensory and dorsal root ganglion neurons, and in the retina. We observe a widespread requirement for Rac1 in axon growth and guidance and a cell-autonomous defect in axon growth in Rac1 (-/-) motor neurons in culture. Neuronal death, presumably a secondary consequence of the axon growth and/or guidance defects, was observed in multiple locations. Following deletion of Rac1 in the forebrain, thalamocortical axons were misrouted inferiorly, with the majority projecting to the contralateral thalamus and a minority projecting ipsilaterally to the ventral cortex, a pattern of misrouting that is indistinguishable from the pattern previously observed in Frizzled3 (-/-) and Celsr3 (-/-) forebrains. In the limbs, motor-neuron-specific deletion of Rac1 produced a distinctive stalling of axons within the dorsal nerve of the hindlimb but a much milder loss of axons in the ventral hindlimb and forelimb nerves, a pattern that is virtually identical to the one previously observed in Frizzled3 (-/-) limbs.The similarities in axon growth and guidance phenotypes caused by Rac1, Frizzled3, and Celsr3 loss-of-function mutations suggest a mechanistic connection between tissue polarity/planar cell polarity signaling and Rac1-dependent cytoskeletal regulation. | | | 26395878
 |
Immunohistochemical demonstration of specific antigens in the human brain fixed in zinc-ethanol-formaldehyde.
Korzhevskii, DE; Sukhorukova, EG; Kirik, OV; Grigorev, IP
European journal of histochemistry : EJH
59
2530
2015
Show Abstract
Tissue fixation is critical for immunohistochemistry. Recently, we developed a zinc-ethanol-formalin fixative (ZEF), and the present study was aimed to assess the applicability of the ZEF for the human brain histology and immunohistochemistry and to evaluate the detectability of different antigens in the human brain fixed with ZEF. In total, 11 antigens were tested, including NeuN, neuron-specific enolase, GFAP, Iba-1, calbindin, calretinin, choline acetyltransferase, glutamic acid decarboxylase (GAD65), tyrosine hydroxylase, synaptophysin, and α-tubulin. The obtained data show that: i) the ZEF has potential for use in general histological practice, where detailed characterization of human brain morphology is needed; ii) the antigens tested are well-preserved in the human brain specimens fixed in the ZEF. | | | 26428887
 |
Forebrain deletion of the dystonia protein torsinA causes dystonic-like movements and loss of striatal cholinergic neurons.
Pappas, SS; Darr, K; Holley, SM; Cepeda, C; Mabrouk, OS; Wong, JM; LeWitt, TM; Paudel, R; Houlden, H; Kennedy, RT; Levine, MS; Dauer, WT
eLife
4
e08352
2015
Show Abstract
Striatal dysfunction plays an important role in dystonia, but the striatal cell types that contribute to abnormal movements are poorly defined. We demonstrate that conditional deletion of the DYT1 dystonia protein torsinA in embryonic progenitors of forebrain cholinergic and GABAergic neurons causes dystonic-like twisting movements that emerge during juvenile CNS maturation. The onset of these movements coincides with selective degeneration of dorsal striatal large cholinergic interneurons (LCI), and surviving LCI exhibit morphological, electrophysiological, and connectivity abnormalities. Consistent with the importance of this LCI pathology, murine dystonic-like movements are reduced significantly with an antimuscarinic agent used clinically, and we identify cholinergic abnormalities in postmortem striatal tissue from DYT1 dystonia patients. These findings demonstrate that dorsal LCI have a unique requirement for torsinA function during striatal maturation, and link abnormalities of these cells to dystonic-like movements in an overtly symptomatic animal model. | | | 26052670
 |
Human Embryonic Stem Cell-Derived Progenitors Assist Functional Sensory Axon Regeneration after Dorsal Root Avulsion Injury.
Hoeber, J; Trolle, C; Konig, N; Du, Z; Gallo, A; Hermans, E; Aldskogius, H; Shortland, P; Zhang, SC; Deumens, R; Kozlova, EN
Scientific reports
5
10666
2015
Show Abstract
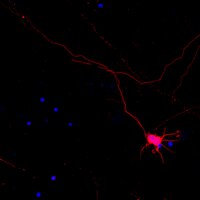
Dorsal root avulsion results in permanent impairment of sensory functions due to disconnection between the peripheral and central nervous system. Improved strategies are therefore needed to reconnect injured sensory neurons with their spinal cord targets in order to achieve functional repair after brachial and lumbosacral plexus avulsion injuries. Here, we show that sensory functions can be restored in the adult mouse if avulsed sensory fibers are bridged with the spinal cord by human neural progenitor (hNP) transplants. Responses to peripheral mechanical sensory stimulation were significantly improved in transplanted animals. Transganglionic tracing showed host sensory axons only in the spinal cord dorsal horn of treated animals. Immunohistochemical analysis confirmed that sensory fibers had grown through the bridge and showed robust survival and differentiation of the transplants. Section of the repaired dorsal roots distal to the transplant completely abolished the behavioral improvement. This demonstrates that hNP transplants promote recovery of sensorimotor functions after dorsal root avulsion, and that these effects are mediated by spinal ingrowth of host sensory axons. These results provide a rationale for the development of novel stem cell-based strategies for functionally useful bridging of the peripheral and central nervous system. | Immunohistochemistry | | 26053681
 |
Restoring the ON Switch in Blind Retinas: Opto-mGluR6, a Next-Generation, Cell-Tailored Optogenetic Tool.
van Wyk, M; Pielecka-Fortuna, J; Löwel, S; Kleinlogel, S
PLoS biology
13
e1002143
2015
Show Abstract
Photoreceptor degeneration is one of the most prevalent causes of blindness. Despite photoreceptor loss, the inner retina and central visual pathways remain intact over an extended time period, which has led to creative optogenetic approaches to restore light sensitivity in the surviving inner retina. The major drawbacks of all optogenetic tools recently developed and tested in mouse models are their low light sensitivity and lack of physiological compatibility. Here we introduce a next-generation optogenetic tool, Opto-mGluR6, designed for retinal ON-bipolar cells, which overcomes these limitations. We show that Opto-mGluR6, a chimeric protein consisting of the intracellular domains of the ON-bipolar cell-specific metabotropic glutamate receptor mGluR6 and the light-sensing domains of melanopsin, reliably recovers vision at the retinal, cortical, and behavioral levels under moderate daylight illumination. | Immunoblotting (Western) | | 25950461
 |